Abstract
Background: The T-cell immunoglobulin and ITIM domain (TIGIT) is highly coexpressed with programmed cell death 1 (PD-1) on both CD4 and CD8 T cells in cancers. Blockade of TIGIT with vibostolimab (MK-7684) has demonstrated antitumor activity in multiple preclinical tumor models. Inhibition of the PD-1 pathway with pembrolizumab has demonstrated efficacy and safety in several hematologic malignancies. Enhanced antitumor activity with anti-TIGIT and anti-PD-L1 combination therapy has been seen in a preclinical model. A multicohort, open-label, phase 2 study will evaluate the safety and efficacy of MK-7684A, a coformulation of vibostolimab and pembrolizumab, in patients with relapsed or refractory (r/r) hematologic malignancies.
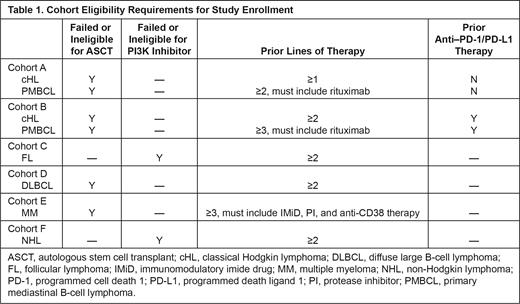
Study Design and Methods: This two-part phase 2 study includes a signal-finding phase (part 1) with the option for a dose-expansion phase (part 2), depending on the observed risk-benefit profile from part 1. Patients must have confirmed diagnosis of r/r disease, including classical Hodgkin lymphoma (cHL; cohort A or B), primary mediastinal B-cell lymphoma (PMBCL; cohort A or B), follicular lymphoma (FL; cohort C), diffuse large B-cell lymphoma (DLBCL; cohort D), multiple myeloma (MM; cohort E), or non-Hodgkin lymphoma (NHL; cohort F) (Table 1). Three hundred thirty participants are expected to enroll in part 1 (signal finding) and part 2 (cohort expansion). In part 1, patients in cohorts A-F will receive MK-7684A (vibostolimab 200 mg + pembrolizumab 200 mg) by IV infusion every 3 weeks (Q3W) for up to 35 cycles. Patients will continue treatment until investigator-determined PD, start of new anticancer treatment, documented complete response, or completion of maximum 35 cycles of treatment. Safety data from the first 12 treated patients from any cohort will be collected. Dose-limiting toxicities (DLTs) will be monitored for the first 6 weeks of continuous treatment. If ≥5 of the 12 patients experience DLTs during this period, enrollment may be discontinued. After the DLT review period, safety data will be collected every 6 months for all enrolled patients. Efficacy assessments will be performed Q3W, with the initial tumor imaging performed ≤28 days after enrollment and repeated at 12 weeks (cohorts A-D, F) or 4 weeks (cohort E) after enrollment. Adverse events (AEs) will be monitored throughout the study, and severity will be graded according to the guidelines outlined in NCI CTCAE version 5.0. Each patient will be monitored for AEs and serious AEs for 30 day and 90 days, respectively, after discontinuation of study intervention. The primary end point for part 1 is the number and proportion of patients with DLTs, AEs, and discontinuations from study treatment due to AEs. Safety and tolerability will be assessed by clinical review. Secondary end points include investigator-assessed objective response rate, investigator-assessed duration of response, disease control rate, and pharmacokinetic end points. Overall survival, progress-free survival, health-related quality of life assessments, and molecular assessments will be exploratory. In the part 2 dose-expansion phase, the plan is to enroll approximately 120 patients from cohorts B-E to receive up to 35 cycles of MK-7684A Q3W; 30 additional patients may be enrolled in a cohort expansion (cohort G) to receive vibostolimab monotherapy Q3W for up to 35 cycles. Cohort G will include patients with cHL or PMBCL with PD after ≥2 or ≥3 prior therapies, FL after ≥2 prior therapies, DLBCL after ≥2 prior therapies, and/or MM after ≥3 prior therapies.
Yusuf: Merck & Co., Inc.: Current Employment. Jemielita: Merck & Co., Inc.: Current Employment, Other: Current Stockholder. Marinello: Merck & Co., Inc.: Current Employment, Other: Current Stockholder.